Neonatal hyperbilirubinemia is one of the most commonly dealt with conditions in pediatrics as all newborns start life with deficits in bilirubin metabolism that require at minimum a bilirubin check prior to their discharge from newborn nursery. Hyperbilirubinemia in infants ≥35 weeks gestation is defined as a total bilirubin >95th percentile for age (in hours of life) and gestation. Hyperbilirubinemia with a total bilirubin >25mg/dL is associated with an increased risk of bilirubin crossing the blood-brain barrier and binding to brain tissue which can cause neurologic dysfunction. The degree to which each individual newborn is affected by hyperbilirubinemia varies based on multiple factors that will be discussed below.
Bilirubin is a product of heme catabolism. Heme is broken down via two steps, first via heme oxidase into the byproducts CO2 and biliverdin. The biliverdin is then broken down by biliverdin reductase into unconjugated bilirubin. Circulating bilirubin is bound by albumin and taken to the liver where it is conjugated by UDP-Glucuronosyltransferase (UGT). Conjugated bilirubin is more water soluble and thus can be excreted as bile into the intestinal lumen and subsequently stooled out by the baby. There is some excretion through the urine, however this is minimal.
Pathophysiology
The most common cause of pathologic indirect hyperbilirubinemia is increased bilirubin production due to hemolytic disease processes that include the following:
- Isoimmune-mediated hemolysis (e.g., ABO or Rh(D) incompatibility)
- Inherited red blood cell membrane defects (e.g., hereditary spherocytosis and elliptocytosis).
- Erythrocyte enzymatic defects (e.g., glucose-6-phosphate dehydrogenase deficiency, pyruvate kinase deficiency, and congenital erythropoietic porphyria).
- Sepsis: likely due increased oxidative stress.
- Polycythemia (infants of diabetic mothers) or sequestration of blood within a closed space (cephalohematoma or caput or bruising).
Inherited defects in the gene that encodes UGT1A1, which catalyzes the conjugation of bilirubin with glucuronic acid. These conditions usually are identified by metabolic screening programs. However, infants may develop severe jaundice before screening results become available.
- Crigler-Najjar syndrome type I: autosomal recessive. The most severe form. UGT activity is absent and severe hyperbilirubinemia develops in the first 2 to 3 days after birth.
- Crigler-Najjar syndrome type II: Usually autosomal recessive but can be autosomal dominant. Less severe. UGT activity is low but detectable. Often responds to phenobarbital treatment.
- Gilbert syndrome: the most common. It results from a reduced production of UGT. Breast milk jaundice during the second week after birth may be due to the concurrent neonatal manifestation of Gilbert syndrome, however later in life flares can be caused by stress/illness.
- Breastfeeding jaundice:
- Also called breastfeeding failure jaundice or suboptimal intake jaundice in order to emphasize that this type of neonatal jaundice is not caused by breastfeeding per se.
- Typically presents in the first week of life with weight loss and dehydration. Hypernatremia, hypophosphatemia, and acidosis are possible.
- The primary mechanism for the increased likelihood of kernicterus and jaundice with breast versus formula feeding is the failure to successfully initiate breastfeeding—total bilirubin is only marginally higher in successfully breastfed compared with formula-fed infants.
- Breast milk jaundice:
- Typically occurs days 3–5, peaks at 2 weeks of life, and resolves by 12 weeks of life.
- Generally milder. Affected infants commonly have total bilirubin levels >5mg/dL for several weeks after delivery.
- Unknown cause. The theory is beta-glucuronidase is a substance in breastmilk that de-conjugates intestinal bilirubin, increasing absorption and enterohepatic circulation.
- Although it may not require intervention, it should be monitored to ensure that it remains unconjugated and does not rise to phototherapy levels.
- The recommendation is to continue breastfeeding and treat as needed rather than temporarily switching to formula.
- Impaired intestinal motility caused by functional (magnesium exposure, short segment Hirschprung’s, gastroparesis) or anatomic obstruction (congenital microcolon, intestinal atresias).
Types of jaundice
The low-risk unconjugated (indirect) bilirubinemia that occurs in nearly all newborns.
- Contributing factors:
- Newborns have more red blood cells (hematocrit between 50 to 60%) and fetal red blood cells have a shorter life span (approximately 85 days) than those in adults, which leads to higher turnover rates at large volumes and thus increased bilirubin production at birth.
- Bilirubin clearance is decreased due to the deficiency of the enzyme uridine diphosphogluconurate glucuronosyltransferase (UGT1A1). UGT activity in term infants does not reach adult levels until 14 weeks of age.
- There is an increase in the enterohepatic circulation of bilirubin due to inconsistent stooling patterns in the first few days of life.
- Usually peaks around 72 hr in a term newborn and around five days in preterm and Asian infants, and then self-resolves within the first one to two weeks after birth (usually by the fifth day in Caucasian and African-American infants and by tenth day in Asian infants).
- The following features should increase suspicion got severe hyperbilirubinemia:
- Jaundice in the first 24 hours: Always pathologic (severe hemolysis, underlying genetic abnormality etc).
- Direct bilirubin > 1.0 (if TB <5) or ≥ 20% of total bilirubin.
- Total bilirubin greater than the 95th% for gestation and hours of life
- Rate of rise on serial checks is >0.2mg/dL/hour.
- Persistent jaundice in a term infant after 2 weeks of age.
- Risk factors:
- Prematurity: decreased liver maturity leads to decreased conjugation ability due to lower enzyme levels.
- ABO incompatibility: Increased risk for hemolysis.
- Family history of jaundice requiring phototherapy in a sibling: There is a genetic component to the level of active enzymes in the liver.
- Cephalohematoma or significant bruising: Increased hemolysis that can be more prolonged than what is expected immediately after birth.
- Exclusive breastfeeding with excessive weight loss commonly thought of as breastfeeding jaundice, linked to dehydration and increased enterohepatic circulation due to decreased stool volume/frequency.
- East Asian race: linked to less conjugation enzyme in the liver.
- Polycythemia at birth:
- Seen in infants of diabetic mothers as these babies will have increased metabolic demands in utero (due to higher maternal glucose through the placenta causing fetal insulin to rise, and that insulin rise is linked to increased cortisol and metabolic demand. To increase oxygen carrying capacity in the setting of the increased metabolic demand, the fetal kidneys produce more EPO and thus there is a rise in hematocrit).
- Delayed cord clamping can cause an increase in newborn hematocrit, however when done appropriately is low risk. Delaying clamping past the recommended timeframe or allowing the placenta to stay attached to baby until it spontaneously falls off (lotus births) are higher risk for causing polycythemia in baby.
Hyperbilirubinemia
- Physiologic jaundice.
- Breastfeeding jaundice (first week).
- Breastmilk jaundice (second week).
- Hemolysis: ABO incompatibility, Rh incompatibility.
- RBC enzyme/membrane defects: G6PD deficiency, Pyruvate kinase deficiency, Hereditary spherocytosis, Hereditary elliptocytosis.
- Conjugation defects:
- Gilbert disease.
- Crigler-Najjar syndrome.
- Other:
- Sepsis.
- Galactosemia.
- Pyloric stenosis. Cephalohematoma or bruising.
- Polycythemia.
- Differential based on age:
- First 24hr: Hemolytic disorders (G6PD, spherocytosis, pyruvate kinase deficiency), Infection: TORCH or sepsis.
- 24hr–21d: Physiologic (often disappears after the first week), breastmilk.
- Breastfeeding.
- Sepsis.
- Resolving cephalohematoma.
- Crigler-Najjar Syndrome.
- Hemolytic disorders (including Gilberts).
- >21d at presentation:
- Breastmilk.
- Hypothyroidism.
- Pyloric stenosis.
- Cholestasis.
- Sepsis
- Extrahepatic biliary obstruction:
- Biliary atresia.
- Choledocal cysts.
- Tumors/masses.
- Neonatal sclerosing cholangitis.
- Metabolic/genetic diseases:
- Hypothyroidism.
- Hypopituitarism.
- Galactosemia.
- Alagille syndrome (paucity of bile ducts).
- Alpha-1 antitrypsin deficiency.
- Cystic fibrosis.
- Maternal diabetes.
- Toxins:
- Drugs (aspirin, acetaminophen, rifampin, alcohol, corticosteroids).
- Cholestasis associated with TPN.
- Neonatal hepatitis.
Monitoring
Every newborn gets a bilirubin screen at 24 hours of life (HOL). When able, transcutaneous bilirubin (Tcb) can be used as this does not require poking baby for blood.
Bilirubin is entered into any one of the readily available jaundice calculators: (2022) Hyperbilirubinemia management guidelines BiliTool™, which help guide management on if/when to recheck and whether a serum bilirubin needs to be sent or not.
- When a Tcb is within 3 points of the treatment threshold at which phototherapy would need to be started, a serum bilirubin (Tsb) should be sent.
- When sending a serum bilirubin, it should be sent as a fractional which includes individual results for both direct and indirect bilirubin as the work up/management differs based on which is elevated. The vast majority of newborns will have an elevated indirect bilirubin.
- The following are neurotoxicity risk factors that increase the risk stratification for newborns with hyperbilirubinemia:
- Iso-immune Hemolytic Disease (Coombs positive).
- G6PD deficiency.
- Asphyxia.
- Significant lethargy.
- Temperature instability.
- Sepsis.
- Acidosis.
- Albumin <3.0 g/dL
- Jaundice in the first 24 HOL requires more thorough evaluation, including fractionated Tsb, CBC with differential, Peripheral smear (to evaluate for possible RBC membrane defects, and a type & Coombs if not already done (to determine ABO incompatibility or autoimmune hemolysis).
- The type and Coombs can often be sent off the cord blood, which minimizes the volume of blood needing to be drawn from baby.
- When mothers are O positive, Rh neg, or positive for antibodies (not including those given Rhogam prior to their checks), newborns will have a Coombs sent on the cord blood to automatically check for hemolysis. There is no standard recommendation for how to monitor a baby who is Coombs positive, although the updated hyperbilirubinemia guidelines from 2022 through the AAP do outline a possible strategy.
- Newborns should have an initial bilirubin checked sooner than 24hr of life, but this can be at 4, 6, or 12 hours. Some institutions will automatically get a Tsb with a CBC and reticulocyte count on these babies. However, others will start with a screening Tcb and then only if needing a serum will they get a CBC at that time.
- Newborns who are Coombs positive should have a newborn blood type sent off the cord blood as this is an easy way to evaluate for ABO incompatibility.
- The initial screening Coombs tests are often direct Coombs, which have a lower sensitivity than indirect Coombs for ABO incompatibility and thus nuance must be had in clinical decision making if the clinical picture is concerning for hemolysis even with a negative Coombs.
- Newborns should have an initial bilirubin checked sooner than 24hr of life, but this can be at 4, 6, or 12 hours. Some institutions will automatically get a Tsb with a CBC and reticulocyte count on these babies. However, others will start with a screening Tcb and then only if needing a serum will they get a CBC at that time.
Treatment
Photons emitted by blue to green wavelength of light convert bilirubin into forms that can be excreted by bile/urine without conjugation. This should be done with an overhead light plus a bili blanket.
- Expected total serum bilirubin decline of 2–3 mg/dL within initial 4–6 hours.
- Newborns will have higher insensible losses under the lights due to slight increase in temperature, feeding plan is important. Supporting a family’s decision to breastfeed is important, and when baby is feeding at the breast it is ideal that they do so with the bili blanket still wrapped around them to optimize time getting phototherapy.
- Stop therapy when total bilirubin is at least 2–3 points below the phototherapy threshold when initial phototherapy was initiated. This recommendation is based on the risk of readmission, and thus in newborns who have continued down-trend of bilirubin there can be a shared decision-making conversation with family regarding discharge despite baby not meeting this threshold.
- Rebound bilirubin (turning off the phototherapy and checking a Tsb in 4–12 hours) should be considered in newborns with concern for hemolysis (positive Coombs, ABO incompatibility, persistent bruising or cephalohematoma) or preterm newborns.
- Directly removes bilirubin from circulation. Needs to be done in a high-level NICU.
- Considered when there are signs of encephalopathy.
- Possible complications:
- Thrombocytopenia.
- Coagulopathy.
- GVHD.
- NEC.
- Portal vein thrombosis.
- Electrolyte abnormalities.
- Cardiac arrhythmia.
- These are utilized in the NICU setting in newborns not responding to traditional phototherapy.
- Intravenous immunoglobulin (inhibits hemolysis): In infants with isoimmune hemolytic disease and rising total bilirubin despite intensive phototherapy, IVIG may be administered to avoid the need for exchange transfusion.
- Phenobarbital (increases conjugation and excretion of bilirubin).
- Ursodeoxycholic acid (increases bile flow).
- Metalloporphyrins (inhibits formation of bilirubin).
Potential complications
- Bilirubin is a neurotoxin that can produce irreversible damage via apoptosis and necrosis. The risk dramatically increases at levels ≥25mg/d because of increased permeability of the blood-brain barrier.
- The brain regions most affected are the basal ganglia, hippocampus, and the brainstem nuclei for occulomotor and auditory function.
- The clinical signs of ABE in the neonatal period represent a spectrum. Early signs include high-pitched cry, hypotonia. Worsening signs include lethargy with a poor suck, irritability, and signs of retrocollis or opisthotonus. Advanced signs include bicycling movements of the arms and legs, seizure activity, inconsolable crying, inability to eat, apnea, hypertonicity, weak cry, and coma; Active intervention at this point is unlikely to affect the risk of the development of kernicterus.
- AAP guidelines recommend that infants showing signs of moderate to severe ABE be given an immediate exchange transfusion, even if the total serum bilirubin is falling.
- If suspected in the outpatient setting, infants with symptoms of ABE should be admitted directly to the hospital and NOT be referred to the emergency department where treatment may be delayed.
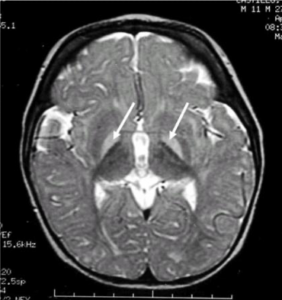 Chronic and usually permanent sequelae of bilirubin neurotoxicity.
Chronic and usually permanent sequelae of bilirubin neurotoxicity.- The major features include:
- Choreoathtoid cerebral palsy (chorea, ballismus, tremor, dystonia).
- Sensorineural hearing loss.
- Gaze abnormalities, especially limitation of upward gaze.
- Dental enamel dysplasia.
- Cognitive function is usually spared.
- Example MRI of the head identifies localization of kernicterus (hyperintense basal ganglia lesions on T2-weighted images).
Prevention
- Initiation of successful feeding is one of the mainstays of preventing hyperbilirubinemia. During the first postpartum week, mothers should nurse whenever the infant shows signs of hunger or when four hours have elapsed since the last feeding—results in 8 to 12 feedings in 24 hours. Problems with breastfeeding should be addressed during initial hospitalization after birth.
- At discharge, a primary care appointment should be scheduled so that the infant-mother dyad is evaluated 24 to 48 hours after discharge, and post-discharge lactation resources provided.
- Supplementation should be considered when the infant has lost more than 7% of birth weight (BW) at 24 HOL, more than 9% of BW at 48 HOL, or more than 10% of BW at 7 days of life or exhibits signs of dehydration. Supplementation can be via expressed breastmilk, donor breastmilk, or formula.
- Neonates should be screened for risk factors, and all infants should have total bilirubin checked by 24 hours of life and at discharge (if >12hr since last check).
Conclusion
Total serum bilirubin levels >1mg/dL occur in almost all term and near-term newborn infants. Infants with severe hyperbilirubinemia are at risk for neurotoxicity, presenting acutely as acute encephalopathy and if inadequately treated can lead to long-term neurologic sequelae or kernicterus. Neonatal jaundice is primarily caused by normal neonatal alterations in bilirubin metabolism including increased bilirubin production, decreased bilirubin clearance, and increased enterohepatic circulation. Hyperbilirubinemia is caused by exaggeration of mechanisms that cause neonatal jaundice or by pathologic conditions that increase bilirubin production, decrease bilirubin clearance, or increase the enterohepatic circulation. Identification of the cause of neonatal hyperbilirubinemia is useful in determining whether therapeutic interventions can prevent severe hyperbilirubinemia.
- Kemper A, et al. Clinical Practice Guideline Revision: Management of Hyperbilirubinemia in the Newborn Infant 35 or More Weeks of Gestation. Pediatrics. August 2022; 150 (3): e2022058859. 10.1542/peds.2022-058859.
Image credits
Unless otherwise noted, images are from Adobe Stock.